Due to the global organ shortage and limited organ donors, thousands of patients are left wanting organs and tissues in cases of severe injuries, illness or genetic conditions. Many of these patients die before transplants are available.
Tissue engineering is an emerging field that works on producing artificial tissue and organ substitutes as permanent solutions to replace or repair damage.
As biomedical engineering researchers, we are developing 3D temporary organ structures — called scaffolds — that may help regenerate damaged tissues and potentially lead to creating artificial organs. These tissues can also be used in various tissue engineering applications, including nerve repair in structures constructed from biomaterials.
Printing tissue
Approximately 22.6 million patients require neurosurgical interventions annually around the world to treat damage to the peripheral nervous system. This damage is primarily caused by traumatic events such as motor vehicle accidents, violence, workplace injuries or difficult births. It is anticipated that the cost of global nerve repair and regeneration will reach more than $400 million by 2025.
Current surgical techniques allow surgeons to realign nerve ends and encourage nerve growth. However, the incidence of recovery in the injured nervous system is not guaranteed, and the return of function is almost never complete.
Animal studies on rats have shown that if an injury destroys more than two centimetres of nerves, the gap cannot be bridged properly and may result in the loss of muscle function or feeling. In this condition, it is important to use a scaffold to bridge two sides of the damaged nerve, specifically in case of large nerve injuries.
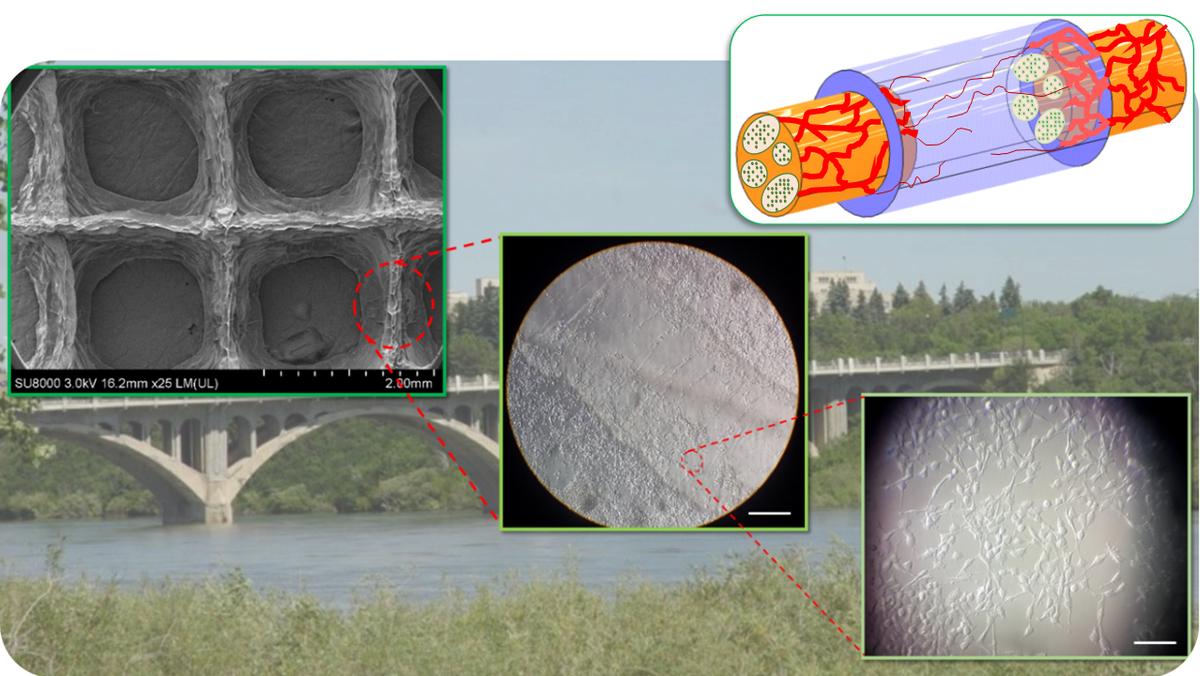
3D bioprinting prints 3D structures layer by layer, similar to 3D printers. Using this technique, our research team created a porous structure made of the patient’s neural cells and a biomaterial to bridge an injured nerve. We used alginate — derived from algae — because the human body does not reject it.
While this technique has not yet been tested in people, once refined, it has the potential to help patients waiting for tissues and organs.
Material challenges
Alginate is a challenging material to work with because it collapses easily during 3D printing. Our research focuses on the development of new techniques to improve its printability.
For nerve repair, alginate has favourable properties for living cells growth and functions, but its poor 3D printability considerably limits its fabrication. It means that alginate flows easily during the printing process, and results in a collapsed structure. We developed a fabrication method where cells are contained within a porous alginate structure that is created with a 3D printer.
Previous research used moulding techniques to create a bulk alginate without a porous structure to improve nerve regeneration; the cells do not like such a solid environment. However, 3D-printing a porous alginate structure is challenging and often impossible.
Our research addresses this issue by printing a porous structure made of alginate layer-by-layer rather than a moulded bulk algiante; such structure has interconnected pores and provides a cell-friendly environment. Cells can easily communicate with each other and start the regeneration while the 3D-printed alginate provides a temporary support for them.
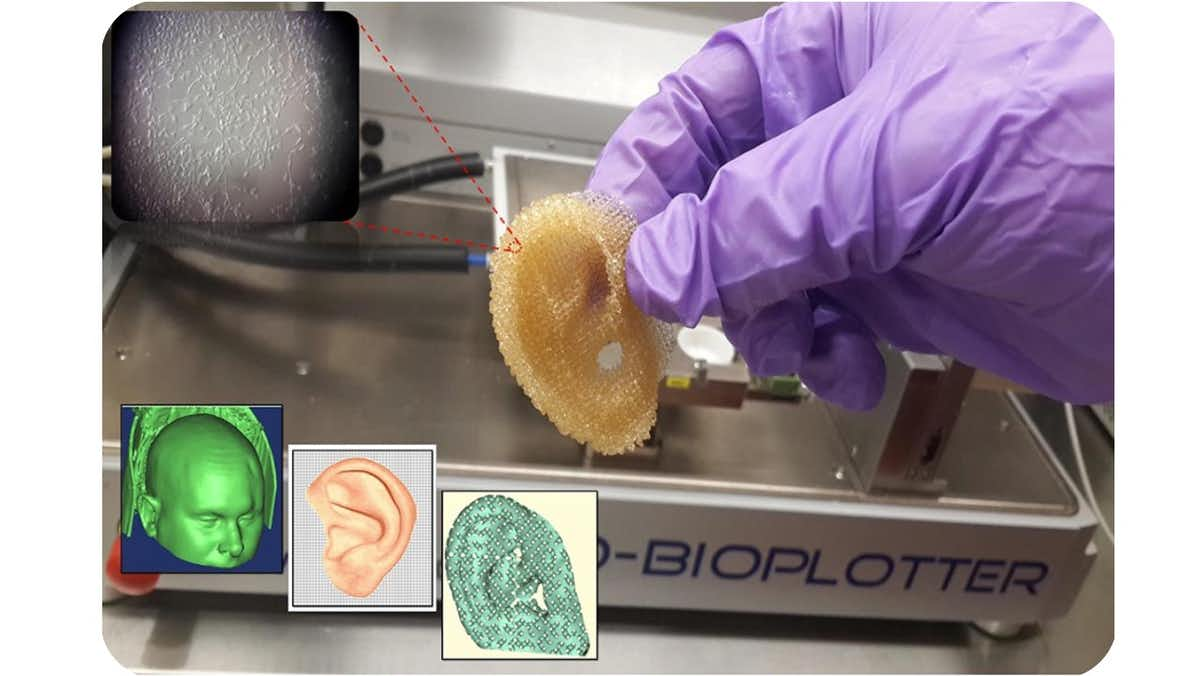
Researchers are going towards the implementation of 3D-printed structures for patients who suffer from nerve injuries as well as other injuries.
After the fabricated alginate structure is implanted in a patient, the big question is if it have enough mechanical stability to tolerate the forces applied by tissues in the body. We developed a novel numerical model to predict the mechanical behaviour of alginate structures.
Our studies will help to understand cell response, which is the main factor to take into account when evaluating the success of the alginate structures.
- is Design Engineer & Research Assistant, University of Saskatchewan
- This article first appeared on The Conversation




